
The third and the fourth definitions that reference the kinetic energy of particles and the ability of a substance to transfer heat are scientifically accurate. But our understanding of temperature is not furthered by such definitions. Any of various standardized numerical measures of this ability, such as the Kelvin, Fahrenheit, and Celsius scale.įor certain, we are comfortable with the first two definitions - the degree or measure of how hot or cold an object is.A measure of the ability of a substance, or more generally of any physical system, to transfer heat energy to another physical system.A measure of the average kinetic energy of the particles in a sample of matter, expressed in terms of units or degrees designated on a standard scale.A measure of the warmth or coldness of an object or substance with reference to some standard value.The degree of hotness or coldness of a body or environment.
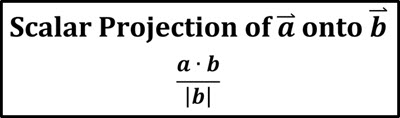
At the risk of doing a belly flop in the pool of enlightenment, I will list some of those definitions here: where I find definitions that vary from the simple-yet-not-too-enlightening to the too-complex-to-be-enlightening. But it is at this point that I'm stumped. It seems that a tutorial page exploring the topic of temperature and thermometers should begin with a simple definition of temperature. But we will likely all agree that we possess built-in thermometers for making qualitative judgments about relative temperatures.ĭespite our built-in feel for temperature, it remains one of those concepts in science that is difficult to define. We may not always agree on whether the room temperature is too hot or too cold or just right. We certainly have a good feel for how one temperature is qualitatively different than another temperature. The water in the shower or bathtub feels hot or cold or warm. We even have a shared language that we use to qualitatively describe temperature. We all have a feel for what temperature is.
